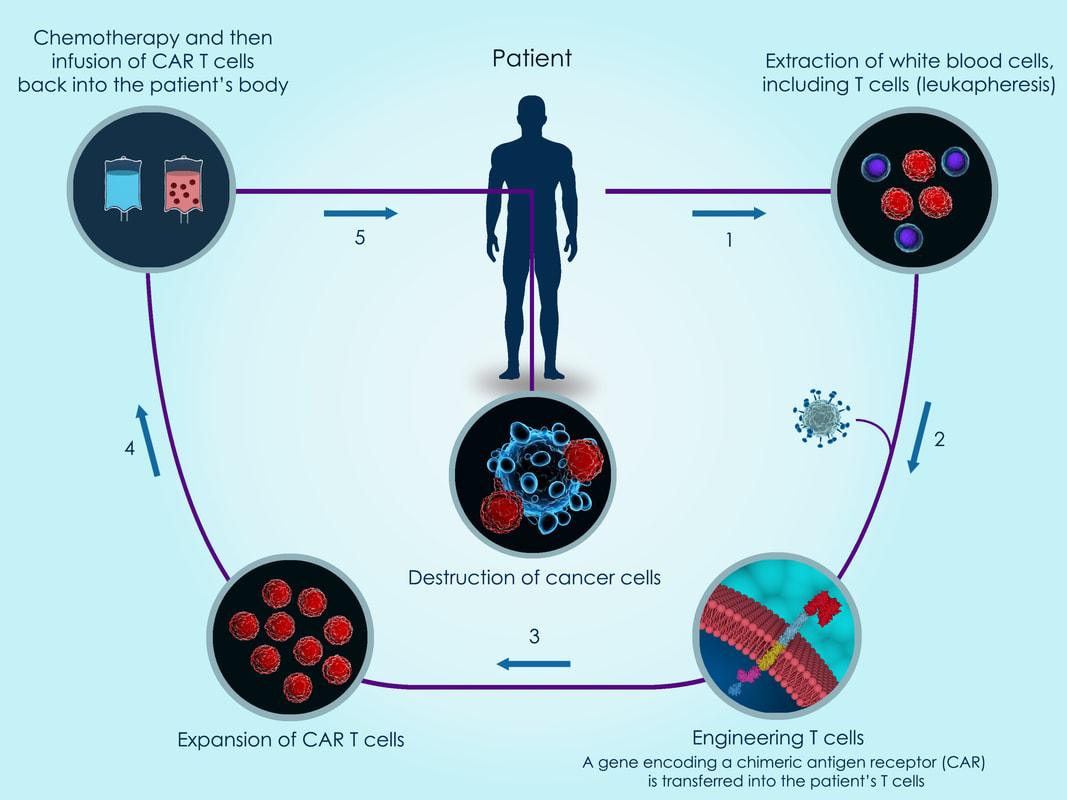
CAR T Cell Therapies
A T cell is a type of lymphocyte, which develops in the thymus gland and plays a central role in the immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell receptor on the cell surface. These immune cells originate as precursor cells, derived from bone marrow, and develop into several distinct types of T cells once they have migrated to the thymus gland. T cell differentiation continues even after they have left the thymus.
Monitoring and Follow Up
As part of a manufacturer's certification of a CAR T cell treatment center, clinics may be required to implement a program to train healthcare staff who prescribe, dispense, or administer CAR T cell therapy about how to monitor, manage, and educate patients on adverse events.
Serious toxicities are known to occur with CAR T cell therapies. These toxicities may require immediate medical attention and may sometimes result in death. They occur in the first few days to weeks after CAR T cell treatment.4,9
Two of the most serious adverse events are Cytokine-release syndrome (CRS) and Neurological toxicity (NT)9:
These are not all of the side effects associated with CAR T cell therapies, and the side effects are different from product to product.
Further information on adverse event monitoring and management can be obtained from CAR T cell therapy manufacturers or study investigators.
Serious toxicities are known to occur with CAR T cell therapies. These toxicities may require immediate medical attention and may sometimes result in death. They occur in the first few days to weeks after CAR T cell treatment.4,9
Two of the most serious adverse events are Cytokine-release syndrome (CRS) and Neurological toxicity (NT)9:
- • CRS symptoms can be progressive, must include fever at the onset and may include hypotension, capillary leak (hypoxia), and end organ dysfunction9
- • NT symptoms or signs can be progressive and may include aphasia, altered level of consciousness, impairment of cognitive skills, motor weakness, seizures, and cerebral edema9
- • Serious Infections
- • Prolonged Cytopenias
- • Hypogammaglobulinemia
These are not all of the side effects associated with CAR T cell therapies, and the side effects are different from product to product.
Further information on adverse event monitoring and management can be obtained from CAR T cell therapy manufacturers or study investigators.
- Celgene
Adverse Reactions
Cytokine-release syndrome (CRS)
Neurological toxicity (NT)9